
Chemistry, 15.05.2021 14:00, mathman783
Calculate the amount of 0.1 M base needed to neutralize 10,000 liters of pH 6.0 water. The base is Sodium Hydroxide (NaOH) Please show work!

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:30, leilanimontes714
Asample of silver (with work function ? = 4.52 ev) is exposed to an ultraviolet light source (? = 200 nm), which results in the ejection of photoelectrons. what changes will be observed if: silver is replaced with copper (? = 5.10 ev) more photoelectrons ejected no photoelectrons are emitted fewer photoelectrons ejected more energetic photoelectrons (on average) less energetic photoelectrons (on average)
Answers: 3

Chemistry, 22.06.2019 02:30, fordkenae
24 points and brainliest to anyone who can answer under 10 minutes with best ! the table below shows the role of different substances during photosynthesis. substance role during photosynthesis glucose stores chemical energy water combines with glucose to form carbon dioxide chlorophyll traps sunlight which of the following statements would correct one of the roles listed in the table? glucose combines with carbon to form water. chlorophyll reacts with light to produce carbon dioxide. water combines with carbon dioxide during photosynthesis. chlorophyll stores chemical energy needed for photosynthesis.
Answers: 1


Chemistry, 22.06.2019 19:30, youngdelvin123
Acetylene gas c2h2 undergoes combustion to produce carbon dioxide and water vapor how many grams of water are produced by the same amount of c2h2?
Answers: 2
Do you know the correct answer?
Calculate the amount of 0.1 M base needed to neutralize 10,000 liters of pH 6.0 water.
The base is...
Questions in other subjects:


Social Studies, 14.10.2021 23:20

Mathematics, 14.10.2021 23:20


Health, 14.10.2021 23:20

Biology, 14.10.2021 23:20

Social Studies, 14.10.2021 23:20


Mathematics, 14.10.2021 23:20


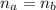
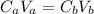
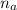 : is the number of moles of the acid
: is the number of moles of the acid 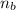 : is the number of moles of the base
: is the number of moles of the base 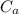 : is the concentration of the acid
: is the concentration of the acid  : is the concentration of the base = 0.1 M
: is the concentration of the base = 0.1 M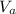 : is the volume of the acid = 10000 L
: is the volume of the acid = 10000 L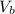 : is the volume of the base =?
: is the volume of the base =?![pH = -log([H^{+}])](/tpl/images/1326/0955/0d4b9.png)
![[H^{+}] = 10^{-pH} = 10^{-6} M](/tpl/images/1326/0955/c9161.png)





