
Chemistry, 14.05.2021 23:40, amarshall90
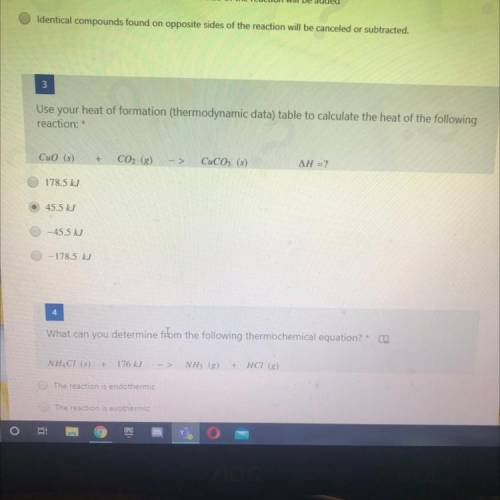
Use your heat of formation (thermodynamic data) table to calculate the heat of the following
reaction:
CuO (5)+CO2 (8)->CuCO2 (s)
AH = ?
178.5 kJ
45.5 kJ
-45.5 kJ
-178.5 kJ

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:10, coralstoner6793
Which of these will change if the air in aclosed bottle is heated? abcdthe mass of the airthe composition of the airthe air pressure in the bottlethe number of air molecules in the bottle
Answers: 3

Chemistry, 22.06.2019 03:30, jabper5522
At a temperature of 393 k, the temperature of a sample of nitrogen is 1.07 atm what will the pressure be at a temperature of 478 k
Answers: 1

Chemistry, 22.06.2019 07:30, reaperqueen21
Compare and contrast the bohr model and the electron cloud models of the atom.
Answers: 1
Do you know the correct answer?
Use your heat of formation (thermodynamic data) table to calculate the heat of the following
reacti...
Questions in other subjects:



Mathematics, 19.02.2021 14:00

Chemistry, 19.02.2021 14:00

Mathematics, 19.02.2021 14:00

Mathematics, 19.02.2021 14:00

History, 19.02.2021 14:00

English, 19.02.2021 14:00


Mathematics, 19.02.2021 14:00






