
Chemistry, 14.05.2021 21:30, dpazmembreno
If I start with 32g S and 56 g Fe, how many grams of product (FeS) will I have? (Assuming that all of the reactants are used up.)
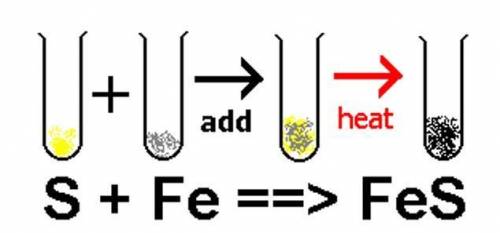

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:00, sabahfayaskhan
Which of the following statements is true? question 4 options: nuclear decay rates vary with the conditions of the reaction, but chemical reaction rates do not. chemical reaction rates vary with the conditions of the reaction, but nuclear decay rates do not. neither chemical reaction rates nor nuclear decay rates vary with the conditions of the reaction. both chemical reaction rates and nuclear decay rates vary with the conditions of the reaction.
Answers: 1

Chemistry, 21.06.2019 19:30, umimgoingtofail
Motivation cannot be developed with practice; a person either possesses it or they do not.
Answers: 1

Chemistry, 22.06.2019 07:30, reaperqueen21
Compare and contrast the bohr model and the electron cloud models of the atom.
Answers: 1

Chemistry, 22.06.2019 10:00, Cythina2007
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1
Do you know the correct answer?
If I start with 32g S and 56 g Fe, how many grams of product (FeS) will I have? (Assuming that all o...
Questions in other subjects:

Spanish, 10.07.2019 05:20

English, 10.07.2019 05:20

Mathematics, 10.07.2019 05:20













