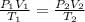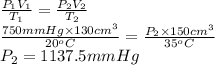130 cm of a gas at 20°C exerts a pressure of
750 mm Hg. Calculate its pressure if its volume
i...

Chemistry, 13.05.2021 17:00, bvolleyball9
130 cm of a gas at 20°C exerts a pressure of
750 mm Hg. Calculate its pressure if its volume
is increased to 150 cm3 at 35 °C.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, andaws21
This large tectonic plate is bounded on three sides by whats know as the ring of fire. what is the name of this tectonic plate? a) pacific plate b) eurasian plate c) north american plate d) indo- australian plate plz it's science but there's no option for science so i picked chemistry
Answers: 2

Chemistry, 22.06.2019 12:00, Unknowndragon42
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2

Chemistry, 22.06.2019 22:30, SavageKidKobe
Rank the four gases (air, exhaled air, gas produced from from decomposition of h2o2, gas from decomposition of nahco3) in order of increasing concentration of co2
Answers: 1

Chemistry, 22.06.2019 23:00, hailey5campbelp7d1c0
In which region is the substance in both the solid phase and the liquid phase? 1 2. 3 4 mark this and return save and exit next
Answers: 2
Do you know the correct answer?
Questions in other subjects:

Social Studies, 15.12.2020 01:00

Mathematics, 15.12.2020 01:00



English, 15.12.2020 01:00


History, 15.12.2020 01:00

Mathematics, 15.12.2020 01:00

History, 15.12.2020 01:00

Mathematics, 15.12.2020 01:00


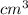 at 35 °C
at 35 °C = 750 mm Hg,
= 750 mm Hg,  ,
, 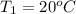
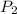 = ?,
= ?,  ,
,