
Chemistry, 12.05.2021 19:30, pamdhuber4224
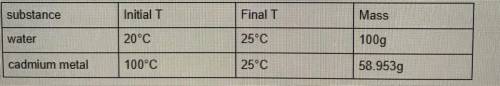
Data in picture
1. First Calculate the heat gained by the water: must show all your work
Examine the result: Is the change positive or negative? Is it endothermic or exothermic? Did the water gain energy or lose energy?
2. Next calculate the heat lost by the cadmium metal: You must show all your work.
Examine the result: Is the change positive or negative? Is it endothermic or exothermic? Did the cadmium gain energy or lose energy?
3. Now, calculate the specific heat of cadmium: You must show all your work.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 16:50, lilblackbird4
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3


Chemistry, 22.06.2019 20:00, emilyswinge4421
Listenbase your answer to the question on the information below. nuclear radiation is harmful to living cells, particularly to fast-growing cells, such as cancer cells and blood cells. an external beam of the radiation emitted from a radioisotope can be directed on a small area of a person to destroy cancer cells within the body. cobalt-60 is an artificially produced radioisotope that emits gamma rays and beta particles. one hospital keeps a 100.0-gram sample of cobalt-60 in an appropriate, secure storage container for future cancer treatment. which choice represents the correct product for the beta decay of the co-60? fe-60ni-60fe-61ni-61
Answers: 2
Do you know the correct answer?
Data in picture
1. First Calculate the heat gained by the water: must show all your work
Questions in other subjects:

Mathematics, 04.03.2020 23:46





Mathematics, 04.03.2020 23:46


English, 04.03.2020 23:46







