
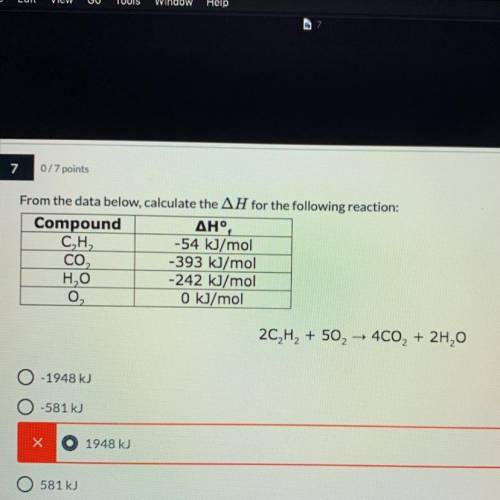
Chemistry, 12.05.2021 09:50, kaieshaweston
From the data below, calculate the AH for the following reaction:(need to know how you got the answer)
2C2H2 + 50, – 400, + 2H,0
A.- 1948 kJ
B.-581 kJ
C.1948 kJ
D.581 kJ

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 11:50, trinityrae4657
Acompound has a molecular weight of 12.124 atomic mass units and the empirical formula c3h40. what is the molecular formula of the compound?
Answers: 3


Chemistry, 22.06.2019 19:50, ellycleland16
Identify the lewis base in this balanced equation: fe3+ h2o fe(h2o)63+
Answers: 1
Do you know the correct answer?
From the data below, calculate the AH for the following reaction:(need to know how you got the answe...
Questions in other subjects:





Chemistry, 25.07.2019 18:00

Chemistry, 25.07.2019 18:00




Mathematics, 25.07.2019 18:00






