The reaction below was carried out in an acidic solution.
[ +103 —>12
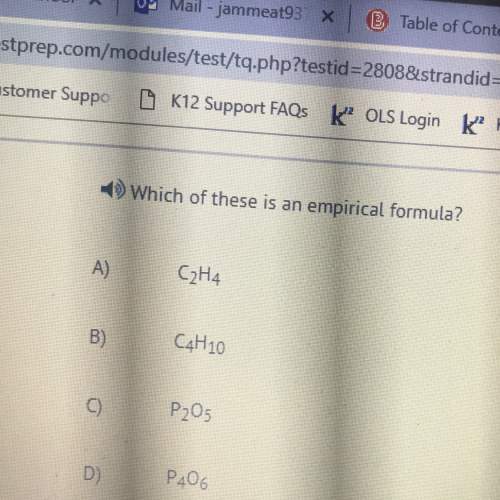
Which statement is...

The reaction below was carried out in an acidic solution.
[ +103 —>12
Which statement is true about this equation?
Although it is unbalanced, it can be balanced by using the half-reaction method.
O It has been balanced by using the half-reaction method.
O It has been balanced by directly using spectator ions.
O Although it is unbalanced, it can be balanced by directly using spectator ions.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:00, miller5452
Acrystal that absorvd water from air is (blank)a. aqueousb. homogenousc. hygroscopicd. efflorescent
Answers: 1

Chemistry, 22.06.2019 12:00, daytonalive83481
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 13:30, annanikherrera
If the concentration of phosphate in the cytosol is 2.0 mm and the concentration of phosphate in the surrounding fluid is 0.1 mm, how could the cell increase the concentration of phosphate in the cytosol? a) passive transportb) diffusionc) active transportd) osmosise) facilitated diffusion
Answers: 3

Chemistry, 22.06.2019 15:00, kamkam5791
Is powdered sports drink ionic or covalent ? 10pts !
Answers: 1
Do you know the correct answer?
Questions in other subjects:



Mathematics, 07.09.2020 02:01




History, 07.09.2020 02:01

Mathematics, 07.09.2020 02:01