
Chemistry, 10.05.2021 19:10, laskew37221
How much energy is required to vaporize 2 kg of copper? Use the table below and this equation: Q = mLvapor-
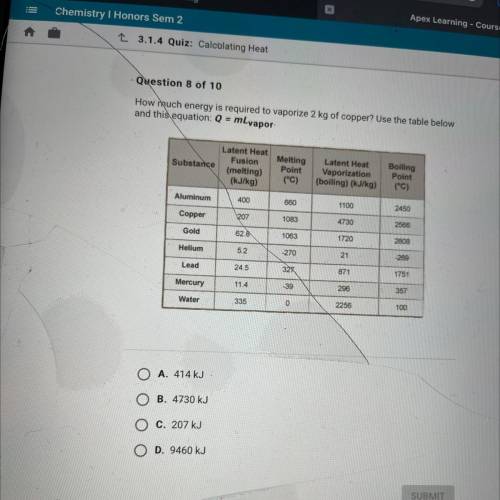

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:40, eamccoy1
Base your answer on the information below and on your knowledge of chemistry. nitrogen dioxide, no2, is a dark brown gas that is used to make nitric acid and to bleach flour. nitrogen dioxide has a boiling point of 294 k at 101.3 kpa. in a rigid cylinder with a movable piston, nitrogen dioxide can be in equilibrium with colorless dinitrogen tetroxide, n2o4. this equilibrium is represented by the equation below. 2no2(g) n2o4(g) + 58kj at standard pressure, compare the strength of intermolecular forces in no2(g) to the strength of intermolecular forces in n2(g).
Answers: 2

Chemistry, 22.06.2019 08:30, audrey1256
Which change in temperature is the smallest? a change of 1 thomson degree a change of 1 kelvin degree a change of 1 fahrenheit degree a change of 1 celsius degree
Answers: 1

Chemistry, 22.06.2019 17:30, latezwardjr15
In a heat of an engine, if 700 j enters the system, and the piston does 400 j of work what is the final internal (thermal) energy of the system if the initial energy is 1,500 j
Answers: 2
Do you know the correct answer?
How much energy is required to vaporize 2 kg of copper? Use the table below and this equation: Q = m...
Questions in other subjects:





Chemistry, 04.07.2019 10:00

English, 04.07.2019 10:00


History, 04.07.2019 10:00

Mathematics, 04.07.2019 10:00

English, 04.07.2019 10:00






