
Chemistry, 10.05.2021 19:00, idbcjcfkxb
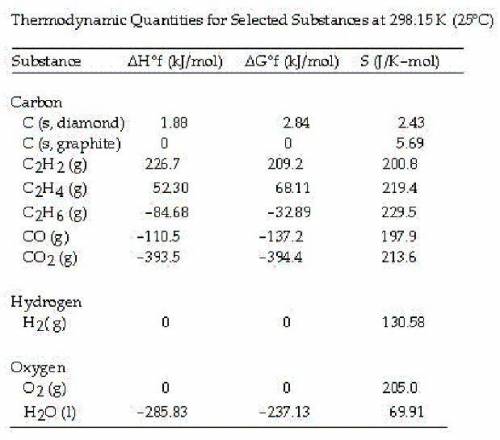
Calculate the value of ΔG° at 25°C for the formation of POCl3 from its constituent elements,
P2 (g) + O2 (g) + 3Cl2 (g) --> 2POCl3 (g)
A. -606.2 kJ/molrxn
B. +1108.7 kJ/molrxn
C. -1108.7 kJ/molrxn
D. +606.2 kJ/molrxn

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 23:00, tovarclaudia055
What does a numerical subscript following an element in a chemical formula mean?
Answers: 1

Chemistry, 23.06.2019 08:00, mackaylabarnes22
Ineed this awnser fast select the correct answer. this chemical equation represents the burning of methane, but the equation is incomplete. what is the missing coefficient in both the reactants and the products? ch4 + → co2 + a. 0 b. 1c. 2d. 3 e. 4
Answers: 3

Do you know the correct answer?
Calculate the value of ΔG° at 25°C for the formation of POCl3 from its constituent elements,
P2 (g...
Questions in other subjects:





Computers and Technology, 12.04.2021 21:20



Mathematics, 12.04.2021 21:20


Mathematics, 12.04.2021 21:20






