
Chemistry, 07.05.2021 03:20, Honeyswish7730
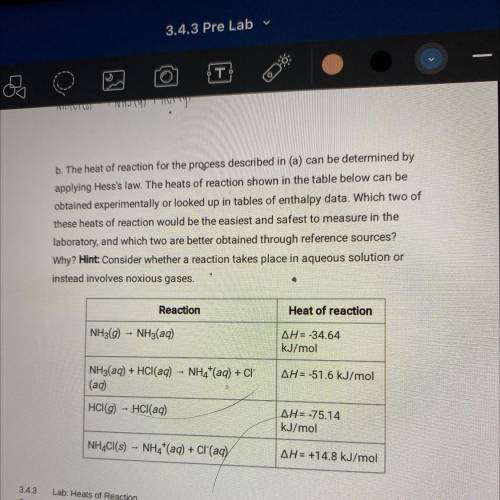
B. The heat of reaction for the process described in (a) can be determined by
applying Hess's law. The heats of reaction shown in the table below can be
obtained experimentally or looked up in tables of enthalpy data. Which two of
these heats of reaction would be the easiest and safest to measure in the
laboratory, and which two are better obtained through reference sources?
Why? Hint: Consider whether a reaction takes place in aqueous solution or
instead involves noxious gases.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, coopera1744
Find the mass in grams of 1.37x1020 particles of h3po4
Answers: 2

Chemistry, 22.06.2019 23:00, ceejay8005
The data below were determined for the reaction shown below. s2o82– + 3i – (aq) → 2so42– + i3– expt. # [s2o82–] (m) [i –] (m) initial rate 1 0.038 0.060 1.4 × 10 – 5 m/s 2 0.076 0.060 2.8 × 10 – 5 m/s 3 0.076 0.030 1.4 × 10 – 5 m/s the rate law for this reaction must be:
Answers: 1

Chemistry, 23.06.2019 02:00, Turtlelover05
Pinene is an unsaturated hydrocarbon found in pine resin. if pinene has m+ = 136 and contains 1 double bond(s) and 2 ring(s); what is its molecular formula? enter the formula in the form ch first, then all other atoms in alphabetical order; do not use subscripts. the formula is case-sensitive
Answers: 3

Chemistry, 23.06.2019 09:00, aaronroberson4940
Weight is a measure of: inertia force matter mass
Answers: 1
Do you know the correct answer?
B. The heat of reaction for the process described in (a) can be determined by
applying Hess's law....
Questions in other subjects:


Mathematics, 31.01.2020 01:44

Mathematics, 31.01.2020 01:44

Mathematics, 31.01.2020 01:44

Mathematics, 31.01.2020 01:44



Mathematics, 31.01.2020 01:44

Mathematics, 31.01.2020 01:44

History, 31.01.2020 01:44






