
Chemistry, 29.04.2021 22:20, amortegaa805
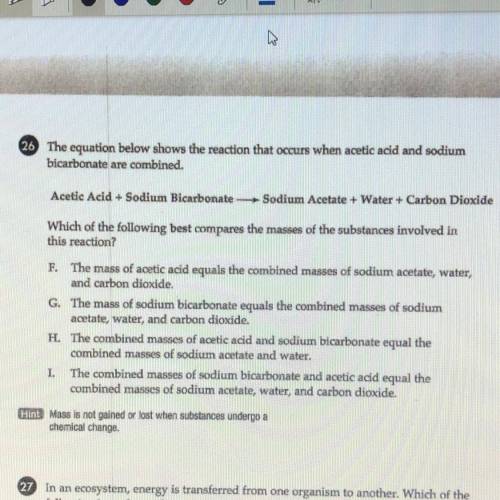
WILL MARK BRAINLIEST PLZ HURRY. 26 The equation below shows the reaction that occurs when acetic acid and sodium
bicarbonate are combined.
Acetic Acid + Sodium Bicarbonate Sodium Acetate + Water + Carbon Dioxide
Which of the following best compares the masses of the substances involved in
this reaction?
F. The mass of acetic acid equals the combined masses of sodium acetate, water,
and carbon dioxide.
G. The mass of sodium bicarbonate equals the combined masses of sodium
acetate, water, and carbon dioxide.
H. The combined masses of acetic acid and sodium bicarbonate equal the
combined masses of sodium acetate and water.
I. The combined masses of sodium bicarbonate and acetic acid equal the
combined masses of sodium acetate, water, and carbon dioxide.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:20, anggar20
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2

Chemistry, 22.06.2019 03:30, jabper5522
At a temperature of 393 k, the temperature of a sample of nitrogen is 1.07 atm what will the pressure be at a temperature of 478 k
Answers: 1


Chemistry, 22.06.2019 14:20, kekecantonxox121
You have a liquid that exhibits diltancy. you want to pour it from a bottle. what should you do to the bottle before pouring
Answers: 1
Do you know the correct answer?
WILL MARK BRAINLIEST PLZ HURRY. 26 The equation below shows the reaction that occurs when acetic aci...
Questions in other subjects:


Mathematics, 23.01.2021 22:20

Physics, 23.01.2021 22:20

Mathematics, 23.01.2021 22:20



History, 23.01.2021 22:20








