
Chemistry, 22.04.2021 01:00, Jadalamanna
calculate the amount of heat energy required to heat up 23.2 grams of ice from -21° C to 56° C ** please show your work**
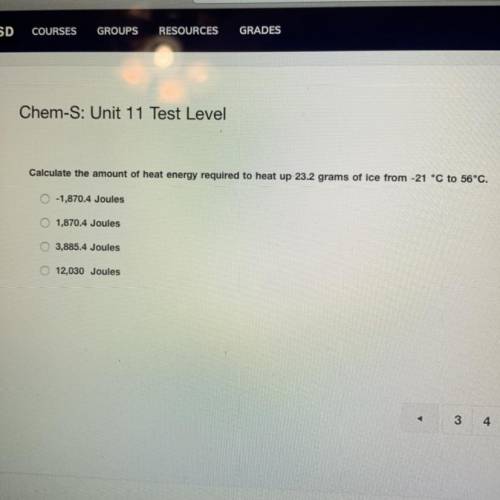

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 20:10, jakhunter354
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3

Chemistry, 23.06.2019 01:00, davelopez979
An unsaturated hydrocarbon is a hydrogen-carbon compound with a. a network solid structure b. single bonds c. single bonds in a branched-chain structure d. double or triple bonds
Answers: 1


Chemistry, 23.06.2019 04:00, hailey200127
What is the volume of 2.5 moles of nitrogen gas (n2)at standard temperature and pressure (stp)?
Answers: 1
Do you know the correct answer?
calculate the amount of heat energy required to heat up 23.2 grams of ice from -21° C to 56° C ** pl...
Questions in other subjects:




History, 27.04.2021 20:20



Mathematics, 27.04.2021 20:20

Social Studies, 27.04.2021 20:20








