
Chemistry, 21.04.2021 23:00, chanahvanya
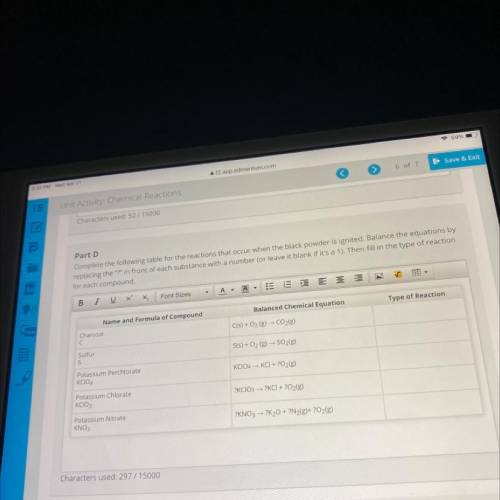
Part D
Complete the following table for the reactions that occur when the black powder is ignited. Balance the equations by
replacing the "?" in front of each substance with a number (or leave it blank if it's a 1). Then fill in the type of reaction
for each compound.
B.
I X
X
Font Sizes
A-
A-
= = 三 三 三
A
Name and Formula of Compound
Balanced Chemical Equation
Type of Reaction
Charcoal
C
C(s) + O2 (g) - CO2(g)
Sulfur
S
S(s) + O2(g) → SO2(g)
Potassium Perchlorate
KCIO4
KCIO4 - KCI + O2(g)
Potassium Chlorate
KCIO3
?KCIO3 → ?KCI + 2O2(g)
Potassium Nitrate
KNO3
?KNO3 → ?K20 + ?N2(g)+ ?O2(g)
Characters used: 297/ 15000

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 20:00, SpiritedAway7087
In vapor-liquid equilibrium in a binary mixture, both components are generally present in both phases. how many degrees of freedom are there for such a system? the reaction between nitrogen and hydrogen to form ammonia occurs in the gas phase. how many degrees of freedom are there for this system? steam and coal react at high temperatures to form hydrogen, carbon monoxide, carbon dioxide, and methane. the following reactions have been suggested as being involved in the chemical transformation:
Answers: 3


Chemistry, 23.06.2019 04:00, Tiredd7838
Which of these are physical changes in matter? check all that apply boiling water a pencil being sharpened exploding dynamite freezing water rotting cheese
Answers: 1
Do you know the correct answer?
Part D
Complete the following table for the reactions that occur when the black powder is ignited....
Questions in other subjects:

Social Studies, 15.12.2020 15:30






History, 15.12.2020 15:30

English, 15.12.2020 15:30







