2.-Agua= H₂O=

Chemistry, 21.04.2021 19:50, vannitling12p4w44f
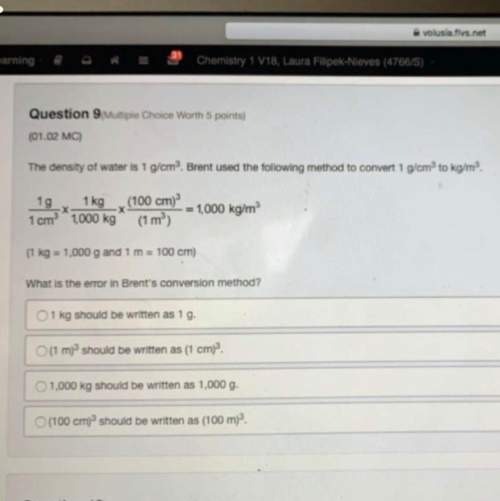
Calcula la masa molar de los siguientes compuestos
1.-Óxido Férrico= Fe₂O₃
2.-Agua= H₂O=
3.-Metano= CH₄=
4.-Hidróxido de sodio= NaOH=
5.-Cloruro de sodio= NaCl

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 23.06.2019 05:00, cpcoolestkid4
C=59(f−32)the equation above shows how temperature f, measured in degrees fahrenheit, relates to a temperature c, measured in degrees celsius. based on the equation, which of the following must be true? a temperature increase of 1 degree fahrenheit is equivalent to a temperature increase of 59 degree celsius. a temperature increase of 1 degree celsius is equivalent to a temperature increase of 1.8 degrees fahrenheit. a temperature increase of 59 degree fahrenheit is equivalent to a temperature increase of 1 degree celsius. a) i onlyb) ii onlyc) iii onlyd) i and ii only
Answers: 1

Chemistry, 23.06.2019 10:30, cjtambasco
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 3.75 mol fe and 8.70 mol nio(oh) react?
Answers: 1


Chemistry, 23.06.2019 22:00, kaylastronaut
The following compounds, listed with their boiling points, are liquid at –10ºc: butane, –0.5ºc; ethanol, 78.3ºc; toluene, 110.6ºc. at –10ºc, which of these liquids would you expect to have the highest vapor pressure? which the lowest? explain. (4 points)
Answers: 1
Do you know the correct answer?
Calcula la masa molar de los siguientes compuestos
1.-Óxido Férrico= Fe₂O₃
2.-Agua= H₂O=
2.-Agua= H₂O=
Questions in other subjects:

Mathematics, 06.05.2020 22:04

Mathematics, 06.05.2020 22:04


Mathematics, 06.05.2020 22:04

Health, 06.05.2020 22:04





Mathematics, 06.05.2020 22:04