
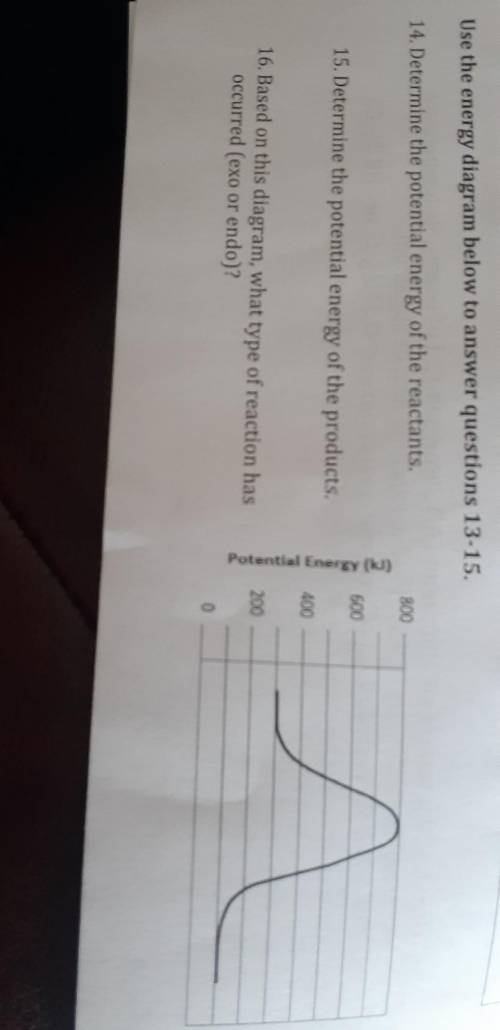
Use the energy diagram below to answer questions 13-15. 14. Determine the potential energy of the reactants. 800 600 15. Determine the potential energy of the products. Potential Energy (kJ) 400 16. Based on this diagram, what type of reaction has occurred (exo or endo)? 200 0

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:10, vapelordcarl69
When 225mg of anthracene, c14h10(s), was burned in a bomb calorimeter the temperature rose by 1.75k. calculate the calorimeter constant. by how much will the temperature rise when 125mg of phenol, c6h5oh(s), is burned in the calorimeter under the same conditions? (δch< (c14h10,s)=–7061 kj mol−1.)
Answers: 3

Chemistry, 22.06.2019 16:50, lilblackbird4
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 19:30, Karinaccccc
How might this scientific phenomena be explained? a paper clip floats on water.
Answers: 1
Do you know the correct answer?
Use the energy diagram below to answer questions 13-15. 14. Determine the potential energy of the re...
Questions in other subjects:


Biology, 13.09.2021 14:00



History, 13.09.2021 14:00

English, 13.09.2021 14:00

Chemistry, 13.09.2021 14:00

Biology, 13.09.2021 14:00

Mathematics, 13.09.2021 14:00






