
Chemistry, 26.09.2019 15:50, devbar3416
Use the periodic to fill in the numbers in the electron configurations shown below. b: 1s2 2sa2pb

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, tifftifftiff5069
You encounter a solution that is acidic and you decide to test it by adding a small amount of a strong acid. the ph lowers slightly but is approximately unchanged, and still remains acidic. what can you say about the solution? a. it is a buffer solution. b. it is not a buffer solution it is a strong acid solution. d. the solution has been neutralized. e. the solution has excess acid present
Answers: 1

Chemistry, 22.06.2019 07:40, sadcase85
22. a flask containing 450 ml of 0.50 m h2so4 was accidentally knocked to the floor. how many grams of nahco, do you need to put on the spill to neutralize the acid according to the following equation: h2so4(aq)+2 nahcos(aq) na, so(aq) +2 h20()+2 co2(g) d) 38 g a) 2.3 g b) 9.5 g c) 19 g
Answers: 1


Chemistry, 22.06.2019 12:30, meghan2529
The melting point of sulfur is 115 °c and its boiling point is 445 °c. what state would sulfur be in at 200 °c?
Answers: 1
Do you know the correct answer?
Use the periodic to fill in the numbers in the electron configurations shown below. b: 1s2 2sa2pb...
Questions in other subjects:

Mathematics, 18.04.2020 01:00




Biology, 18.04.2020 01:00



Mathematics, 18.04.2020 01:00



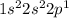 . Boron is a non-metal and belongs to group 13.
. Boron is a non-metal and belongs to group 13.




