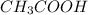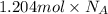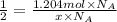Chemistry, 16.09.2019 21:40, jadejordan8888
This is the chemical formula for acetic acid (the chemical that gives the sharp taste to vinegar): ch3co2h an analytical chemist has determined by measurements that there are 1.204 moles of carbon in a sample of acetic acid. how many moles of hydrogen are in the sample? round your answer to 4 significant digits.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:10, shafferakr6
There are 6.022 x 10^23 atoms of hg in 1 mole of hg. the number of atoms in 4.5 moles of hg can be found by multiplying 4.5 by 6.022 x 10^23 a. 2.7 x 10^24 b. 27 x 10^23 c. 2.71 x10^24 d. 27.099 x 10^23
Answers: 3

Chemistry, 21.06.2019 23:00, DarcieMATHlin2589
Write a brief passage describing a neutral atom of nitrogen-14 (n-14). describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. use the periodic table to you. 14 protons and eletrons since its a neutral atom
Answers: 1

Chemistry, 22.06.2019 08:30, aydenmasonc
Which statement describes james chadwick’s discovery.
Answers: 2

Chemistry, 23.06.2019 01:00, breemills9552
What two factors can affect the properties of a hydrocarbon? a. the number of its carbon atoms and the number of single bonds b. the number of its carbon atoms and the arrangement of its atoms c. the arrangement of its atoms and the number of its double bonds
Answers: 1
Do you know the correct answer?
This is the chemical formula for acetic acid (the chemical that gives the sharp taste to vinegar):...
Questions in other subjects:

Social Studies, 16.07.2020 20:01




Mathematics, 16.07.2020 20:01


Mathematics, 16.07.2020 20:01