

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:50, martinez6221
What is the chemical name of the compound na2co3? use the list of polyatomic ions and the periodic table to you answer. a. sodium carbon oxide b. sodium carbonate c. sodium(ll) carbonate d. sodium oxalate
Answers: 1



Chemistry, 22.06.2019 18:00, brisacruz013
Which statement best describes the he properties of iconic compounds ?
Answers: 1
Do you know the correct answer?
What is the henry's law constant for co2 at 20∘c? express your answer to three significant figures...
Questions in other subjects:




Mathematics, 26.07.2020 01:01


Social Studies, 26.07.2020 01:01



History, 26.07.2020 01:01


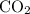 at
at 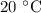 is
is 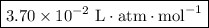 .
.
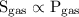 …… (1)
…… (1) 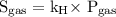 …… (2)
…… (2) 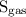 is the solubility of gas.
is the solubility of gas.
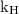 is Henry’s constant.
is Henry’s constant.
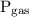 is the pressure of gas.
is the pressure of gas.
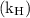 and equation (2) becomes,
and equation (2) becomes,
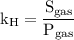 …… (3)
…… (3) 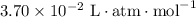 .
.
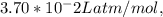 high partial pressure, high solubility.
high partial pressure, high solubility.




