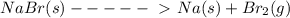Chemistry, 18.09.2019 21:50, Thejollyhellhound20
Solid sodium bromide decomposes into bromine gas and solid sodium . write a balanced chemical equation for this reaction.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:30, robert7248
What is the percent composition of ca(oh)2? 37.7% ca, 53.0% o, and 10.3% h 45.5% ca, 38.2% o, and 16.3% h 54.0% ca, 43.0% o, and 2.7% h 64.7% ca, 27.0% o, and 8.3% h
Answers: 2

Chemistry, 22.06.2019 12:30, fvmousdiana
In france, grapes are 1.95 euros per kilogram. what is the cost of grapes, in dollars per pound, if the exchange rate is 1.14 dollars/euro? (2.6)
Answers: 3


Chemistry, 22.06.2019 18:30, sarahbug56
Which rate indicates the number of children that would be born per woman if she were to live to the end of her child bearing years
Answers: 2
Do you know the correct answer?
Solid sodium bromide decomposes into bromine gas and solid sodium . write a balanced chemical equati...
Questions in other subjects:



Mathematics, 30.11.2020 21:30


Mathematics, 30.11.2020 21:30

English, 30.11.2020 21:30


Physics, 30.11.2020 21:30