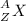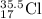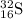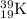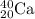Three different atoms or atomic anions with 18 electrons: Cl⁻, K⁺, Ar
Further explanation
The Atomic Number (Z) indicates the number of protons in an atom of an element. If the atom is neutral then the number of protons will be equal to the number of electrons. So the atomic number can also indicate the number of electrons.
So atomic number = number of protons = number of electrons
Mass Number (A) is the sum of protons and neutrons
Mass Number (A) = Number of protons + Number of Neutrons
So that the relationship between atomic numbers and mass numbers can be formulated as follows:
Atomic Number (Z) = Mass Number (A) - Number of Neutrons
In the following element notation,
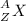
Information
X = symbol of elemental atom
A = mass number
= number of protons + number of neutrons
Z = atomic number
= number of protons = number of electrons, on neutral elements
In an atom, there are levels of energy in the skin and sub skin.
This energy level is expressed in the form of electron configurations.
Charging electrons in the sub skin uses the following sequence:
1s², 2s², 2p⁶, 3s², 3p⁶, 4s², 3d¹⁰, 4p⁶, 5s², 4d¹⁰, 5p⁶, 6s², etc.
The location of the element group in the periodic system is determined by the number of valence electrons that fills the last sub-skin
The element period is determined from the valence shell (the largest main quantum number (n))
Atoms or ionic atoms that have 18 electrons include:
1. Atom: Ar: 1s² 2s² 2p⁶ 3s² 3p⁶2. Atomic ion
a. Cl: 1s² 2s² 2p⁶ 3s² 3p⁵
because it captures 1 more electron to be stable like the noble gas Ar so that the electron configuration has 18 electrons
Cl-: 1s² 2s² 2p⁶ 3s² 3p⁶
b. S: 1s² 2s² 2p⁶ 3s² 3p⁴
catch 2 electrons to be stable
S²⁻: 1s² 2s² 2p⁶ 3s² 3p⁶
c. K: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s¹
release 1 electron to stabilize it
K⁺: 1s² 2s² 2p⁶ 3s² 3p⁶
d. Ca: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s²
release 2 electrons to stabilize
Ca²⁺: 1s² 2s² 2p⁶ 3s² 3p⁶
While the notation of the symbol elements are:
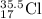
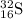
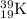
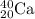
Learn more
element symbol
link
the chemical symbols
link
Identify the group number
link
Keywords: the chemical symbols, electron configuration