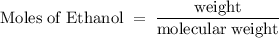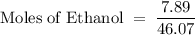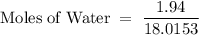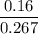Chemistry, 18.09.2019 08:00, davionb556
What is the mole fraction of ethanol when 10.00 ml of pure ethanol is combined with 2.00 ml of water? the volume of 10 ml of the solution must be measured?

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 13:50, amandamac7339
Abeaker with 2.00×102 ml of an acetic acid buffer with a ph of 5.000 is sitting on a benchtop. the total molarity of acid and conjugate base in this buffer is 0.100 m. a student adds 4.70 ml of a 0.360 m hcl solution to the beaker. how much will the ph change? the pka of acetic acid is 4.740.
Answers: 1

Do you know the correct answer?
What is the mole fraction of ethanol when 10.00 ml of pure ethanol is combined with 2.00 ml of water...
Questions in other subjects:



Mathematics, 22.07.2020 03:01

Chemistry, 22.07.2020 03:01

Mathematics, 22.07.2020 03:01


Mathematics, 22.07.2020 03:01




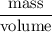
 Volume
Volume