Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:30, Tyrant4life
Amass of 100.0 g of solute is dissolved in water so that 850. ml of a 0.7500 m solution has been prepared. what is the molar mass of the solute?
Answers: 2

Chemistry, 22.06.2019 02:30, brittanysanders
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2

Chemistry, 22.06.2019 05:30, madisonrosamond99
Astudent carefully transfers 30 g of water and 30 g of alcohol in a glass tube, forming two layers and filling the tube completely. after sealing the tube, the student mixes the solutions, and notices a bubble that forms in the tube. what is the mass of the contents in the glass tube after mixing?
Answers: 2

Chemistry, 22.06.2019 12:00, carvajalj2520
Explain what happens at the saturation point when adding salt to water at room temperature.
Answers: 1
Do you know the correct answer?
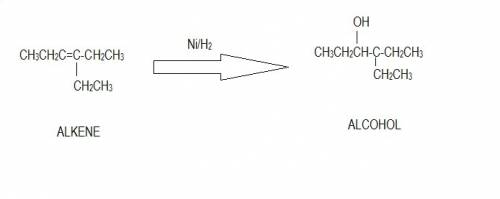
Draw the compound that would produce 4-ethyl-3-hexanol in the presence of a nickel catalyst and hydr...
Questions in other subjects:



Mathematics, 12.06.2020 14:57

Mathematics, 12.06.2020 14:57

English, 12.06.2020 14:57



Social Studies, 12.06.2020 14:57


Physics, 12.06.2020 14:57