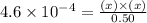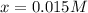Chemistry, 27.09.2019 02:30, myaaa13754
What is the [h3o+] at equilibrium of a 0.50 m weak acid (ha) solution if the ka of the acid is 4.6 × 10−4?

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 23.06.2019 05:00, rosezgomez97
Asolution is made by dissolving 2.3 moles of sodium chloride (nacl) in 0.155 kilograms of water. if the molal boiling point constant for water (kb) is 0.51 °c/m, what would be the boiling point of this solution? show all the steps taken to solve this problem.
Answers: 1

Do you know the correct answer?
What is the [h3o+] at equilibrium of a 0.50 m weak acid (ha) solution if the ka of the acid is 4.6 ×...
Questions in other subjects:

Mathematics, 15.10.2021 14:00


History, 15.10.2021 14:00


English, 15.10.2021 14:00





Biology, 15.10.2021 14:00


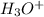 at equilibrium is, 0.015 M
at equilibrium is, 0.015 M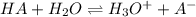
![k_a=\frac{[H_3O^+]\times [A^-]}{[HA]}](/tpl/images/0266/6337/d4f24.png)
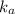 = dissociation constant of weak acid
= dissociation constant of weak acid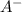 be 'x'
be 'x'