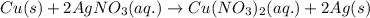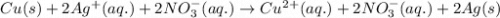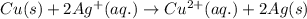Chemistry, 26.09.2019 21:30, cupcake122016
Apiece of copper metal is dipped into an aqueous solution of agno3. which net ionic equation describes the reaction that occurs?

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 07:00, haydjanggg6578
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2

Chemistry, 22.06.2019 09:40, keiracoles
How many grams of aluminum will there be in 98g of al2o3?
Answers: 1

Chemistry, 23.06.2019 00:30, motorxr714
Titration reveals that 11.6 ml of 3.0m sulfuric acid are required to neutralize the sodium hydroxide in 25.00ml of naoh solution. what is the molarity of the naoh solution?
Answers: 1
Do you know the correct answer?
Apiece of copper metal is dipped into an aqueous solution of agno3. which net ionic equation describ...
Questions in other subjects:





History, 18.03.2021 21:00



Mathematics, 18.03.2021 21:00

History, 18.03.2021 21:00

Mathematics, 18.03.2021 21:00