
Chemistry, 09.10.2019 01:30, joshua1255
What mass of carbon dioxide (co2) can be produced from 15.6 g of c6h14 and excess oxygen?

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 17:10, sophiaa23
)benzene and toluene form nearly ideal solutions. consider an equimolar solution of benzene and toluene. at 20 °c the vapour pressures of pure benzene and toluene are 9.9 kpa and 2.9 kpa, respectively. the solution is boiled by reducing the external pressure below the vapour pressure. calculate (i) the pressure when boiling begins, (ii) the composition of each component in the vapour, and (iii) the vapour pressure when only a few drops of liquid remain. assume that the rate of vaporization is low enough for the temperature to remain constant at 20 °c.
Answers: 1

Chemistry, 22.06.2019 20:00, Isaiahtate053
The volume of a single vanadium atom is 9.29×10-24 cm3. what is the volume of a vanadium atom in microliters?
Answers: 3

Chemistry, 23.06.2019 07:30, mazielynn84
If you try to move a piano and are unable to move it, did you perform any work in the scientific sense of the word? yes or no?
Answers: 1
Do you know the correct answer?
What mass of carbon dioxide (co2) can be produced from 15.6 g of c6h14 and excess oxygen?...
Questions in other subjects:


Mathematics, 01.06.2021 19:30

Mathematics, 01.06.2021 19:30

Mathematics, 01.06.2021 19:30

Physics, 01.06.2021 19:30

Mathematics, 01.06.2021 19:30


Mathematics, 01.06.2021 19:30



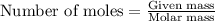 .....(1)
.....(1)
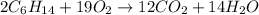
 of carbon dioxide
of carbon dioxide




