
Chemistry, 27.10.2019 14:43, luckilyalexa
Phosphorus trichloride, pcl3, decomposes to form elemental phosphorus and chlorine. the equation is: 4pcl3 → p4 + 6cl2. balance the equation and determine the mass in grams of chlorine that would be formed if 25 grams of pcl3 (molecular mass = 137.32 g/mol) decompose. grams of cl2

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:30, mimithurmond03
A6.10 m nacl can be made by adding [x]g of nacl to a container and making the volume of water up to the 1.00 l line
Answers: 1

Chemistry, 22.06.2019 14:10, roserose3098
16. in a reaction that has reached equilibrium, a. the forward and reverse reactions are occurring at the same rate. b. the reactants and products are in equal concentrations. c. the forward reaction has gone further than the reverse reaction. d. there are equal numbers of atoms on both sides of the equation. e. a, b, and d are correct.
Answers: 2

Chemistry, 22.06.2019 22:40, destineysarah
Covalent bonds generally form when the bonded elements have a difference in electronegativity less than 1.5. subtract the electronegativities for the following pairs of elements and predict whether they form a covalent bond. electronegativity difference of c and c: ionic covalent electronegativity difference of mg and cl: ionic covalent
Answers: 1

Chemistry, 23.06.2019 16:00, kaylaelaine18
When electrons are removed from the outermost shell of a calcium atom, the atom becomes an anion that has a larger radius than the atom. an anion that has a smaller radius than the atom. a cation that has a larger radius than the atom. a cation that has a smaller radius than the atom.
Answers: 3
Do you know the correct answer?
Phosphorus trichloride, pcl3, decomposes to form elemental phosphorus and chlorine. the equation is:...
Questions in other subjects:

Mathematics, 01.12.2021 08:20

Mathematics, 01.12.2021 08:20

Chemistry, 01.12.2021 08:20






English, 01.12.2021 08:30


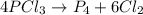
 of particles.
of particles. decompose to give 6 moles of
decompose to give 6 moles of 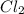
 of
of  of
of 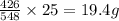 of
of 




