
Chemistry, 04.11.2019 14:31, eprice32315
Use the unbalanced equation nh3 + o2 = no + h2o, what is the mole ratio between nh3 and no

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 11:30, chelseychew32
What is the main reason why some developing countries fear the increase the free trade policies around the world?
Answers: 2

Chemistry, 22.06.2019 11:50, hadwell34
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2

Chemistry, 22.06.2019 12:20, missayers172
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3
Do you know the correct answer?
Use the unbalanced equation nh3 + o2 = no + h2o, what is the mole ratio between nh3 and no...
Questions in other subjects:


History, 28.11.2019 14:31


Mathematics, 28.11.2019 14:31

Mathematics, 28.11.2019 14:31




Computers and Technology, 28.11.2019 14:31

Mathematics, 28.11.2019 14:31


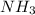 and
and 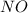 is, 1 : 1
is, 1 : 1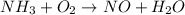
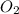 , coefficient 4 is put before
, coefficient 4 is put before 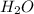 .
.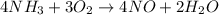
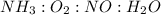 are 4 : 3 : 4 : 2
are 4 : 3 : 4 : 2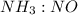 are 4 : 4 or 1 : 1
are 4 : 4 or 1 : 1



