Answer :
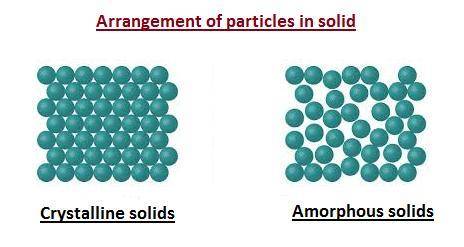
There are two types of solid in terms of arrangement of particles which are crystalline solids and amorphous solids.
Crystalline Solids : It is a type of solids in which the constituent particles of the matter are arranged in the ordered or specific manner. This means that there is a proper arrangement of atoms in solid lattice.
The examples of crystalline solids are, quartz, calcite, sugar, mica, diamonds, etc.
Amorphous Solids : It is a type of solids in which the constituent particles of the matter are arranged in the random or disordered manner. This means that there is no proper arrangement of atoms in solid lattice.
The examples of amorphous solid are, plastics, glass, rubber, metallic glass, polymer, etc.
The arrangement of particles in crystalline solid and amorphous solid are shown below.