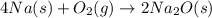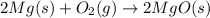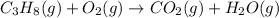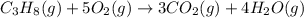Chemistry, 02.12.2019 03:31, itscheesycheedar
Which of the following are balanced equations? check all that apply. a. 4na(s) + o2(g) → 2na2o(s) b. 2mg(s) + o2(g) → 2mgo(s) c. c3h8(g) + o2(g) → co2(g) + h2o(g) d. c3h8(g) + 5o2(g) → 3co2(g) + 4h2o(g

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:00, stephstewart1209
Which of the following observations indicates that there is a small, dense, positively charged part in the center of an atom? some uncharged particles are scattered by a gold foil. all uncharged particles are attracted towards a gold foil. all positively charged particles pass straight through a gold foil. some positively charged particles bounce back from a gold foil.
Answers: 2

Chemistry, 22.06.2019 14:30, clemsongirl5392
Select all that apply. using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 (s) pb+2(aq) + 2cl -(aq). the concentration of the products yield a ksp of 2.1 x 10-2:
Answers: 2

Chemistry, 22.06.2019 21:30, sullivanjakob
Isopropyl alcohol, (ch3)2choh, is a common solvent. determine the percent by mass of hydrogen in isopropyl alcohol. a) 6.71% h b) 13.4% h c) 25.0% h d) 53.3% h
Answers: 1

Chemistry, 22.06.2019 23:50, josie311251
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3
Do you know the correct answer?
Which of the following are balanced equations? check all that apply. a. 4na(s) + o2(g) → 2na2o(s) b...
Questions in other subjects:

Mathematics, 07.01.2020 15:31




History, 07.01.2020 15:31

Mathematics, 07.01.2020 15:31

Mathematics, 07.01.2020 15:31

History, 07.01.2020 15:31