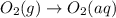o2 (g) --> o2 (aq)

Chemistry, 28.09.2019 14:50, katekayrodriguez10
Given the chemical equation, predict the δs system for this change.
o2 (g) --> o2 (aq)
is it +δs or -δs

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 17:30, kaytonleeb
Take a look at this dandelion. the yellow flower on the right is pollinated and the seeds on the left are transported by
Answers: 2

Chemistry, 22.06.2019 17:30, sheazy3709
The polymer used for the nonstick surface of cooking utensils is 24.0%c and 76%f by mass. what is the empirical formula of this polymer?
Answers: 2

Chemistry, 22.06.2019 20:20, carcon2019
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3
Do you know the correct answer?
Given the chemical equation, predict the δs system for this change.
o2 (g) --> o2 (aq)
o2 (g) --> o2 (aq)
Questions in other subjects:



Biology, 14.07.2019 19:00



Mathematics, 14.07.2019 19:00

English, 14.07.2019 19:00


English, 14.07.2019 19:00

Mathematics, 14.07.2019 19:00


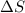 system for this change is, negative
system for this change is, negative 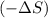 .
.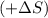 as we are moving from the solid state to liquid state and liquid state to gaseous state and the entropy decreases,
as we are moving from the solid state to liquid state and liquid state to gaseous state and the entropy decreases,