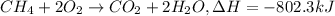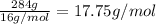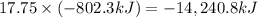Chemistry, 25.10.2019 04:43, walkereddie580
How much heat is produced by the complete combustion of 284 g of ch4? ch4+2o2=co2+2h2o; h=-802.3kj

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:30, emmalybrown
A50.0 ml sample of gas at 20.0 atm of pressure is compressed to 40.0 atm of pressure at constant temperature. what is the new volume? 0.0100 ml 0.325 ml 25.0 ml 100. ml
Answers: 1

Chemistry, 22.06.2019 22:30, teagan56
Gusing the milligrams of ascorbic acid you entered above, the ratio of total sample volume to aliquot volume, and the total milligrams of the vitamin c tablet that you dissolved, calculate the mass of ascorbic acid in the vitamin c tablet for each trial. do this by scaling up to find the amount (mg) of ascorbic acid in your 250 ml flask. enter your calculated mass of ascorbic acid in the vitamin c tablet, for each trial. be sure to enter your calculated mass in the corresponding order that you entered your milligrams of ascorbic acid. the milligrams of ascorbic acid you entered for entry #1 previously should correspond to the mass of ascorbic acid that you enter for entry #1 here.
Answers: 1

Chemistry, 23.06.2019 01:30, heavendl13
How is the solubility of a carbon dioxide gas in water increase?
Answers: 1
Do you know the correct answer?
How much heat is produced by the complete combustion of 284 g of ch4? ch4+2o2=co2+2h2o; h=-802.3kj...
Questions in other subjects:

Mathematics, 19.02.2021 01:00


Physics, 19.02.2021 01:00

History, 19.02.2021 01:00


Computers and Technology, 19.02.2021 01:00

Mathematics, 19.02.2021 01:00

Arts, 19.02.2021 01:00

French, 19.02.2021 01:00

Mathematics, 19.02.2021 01:00