
Avogadro’s law and charles’s law describe a proportionality of the volume of a gas when the pressure is constant. which describes the proportionality that allows these laws to be combined when describing a gas?
volume is directly proportional to temperature (avogadro’s law) and to moles (charles’s law).
volume is inversely proportional to temperature (avogadro’s law) and to moles (charles’s law).
volume is directly proportional to moles (avogadro’s law) and to temperature (charles’s law).
volume is inversely proportional to moles (avogadro’s law) and to temperature (charles’s law).

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:50, bridgetosanders
What are four significant sources of ghgs that come from wostem washington?
Answers: 2

Chemistry, 22.06.2019 12:10, coastieltp58aeg
Building glycogen from glucose molecules is an example of
Answers: 3

Chemistry, 22.06.2019 14:00, MathChic68
Displacement is the slope of a velocity vs. time graph a. true b. false
Answers: 1
Do you know the correct answer?
Avogadro’s law and charles’s law describe a proportionality of the volume of a gas when the pressure...
Questions in other subjects:

Mathematics, 07.06.2021 17:30

Mathematics, 07.06.2021 17:30




Mathematics, 07.06.2021 17:30




English, 07.06.2021 17:30


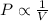 (At constant temperature and number of moles)
(At constant temperature and number of moles)
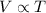 (At constant pressure and number of moles)
(At constant pressure and number of moles)
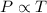 (At constant volume and number of moles)
(At constant volume and number of moles)
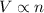 (At constant temperature and pressure)
(At constant temperature and pressure)



