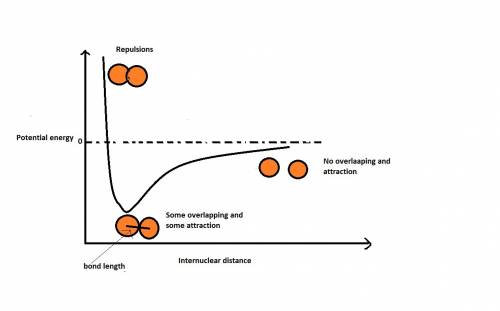
Bond length is the distance between the centers of two bonded atoms. on the potential energy curve, the bond length is the internuclear distance between the two atoms when the potential energy of the system reaches its lowest value. given that the atomic radii of h and cl are 25.0 cm and 100. pm , respectively, predict the bond length of the hcl molecule.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, tddreviews
Match the name of the following compound: mgso4 · h2omagnesium sulfate monohydratemagnesium (ii) sulfate monohydratemagnesium (ii) sulfate hydratemagnesium sulfate hydrate
Answers: 1

Chemistry, 22.06.2019 08:30, omoaye
Identify one disadvantage to each of the following models of electron configuration: -dot structures -arrow and line diagrams -written electron configurations type in your answer below. (answer) -dot structures do not show the distribution of electrons in orbitals and take up a lot of space. -arrow and line diagrams take up a lot of space and make it difficult to count electrons. -written configurations make it easy to lose count of electrons and do not show the distribution of electrons in orbitals.
Answers: 3


Chemistry, 23.06.2019 18:30, zalyndevola
Why must pencil be used to draw on paper in paper chromatography
Answers: 2
Do you know the correct answer?
Bond length is the distance between the centers of two bonded atoms. on the potential energy curve,...
Questions in other subjects:






Chemistry, 16.10.2019 19:00



Computers and Technology, 16.10.2019 19:00