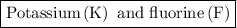 are more likely to form ionic bond.
are more likely to form ionic bond.
Further Explanation:
Chemical compound:
The formation of such compounds involves attraction between different constituent atoms in compound. This interaction between atoms and other constituent particles is termed as chemical bond. Such bonds are formed as result of sharing or transference of electrons from one entity to another.
Covalent compound:
These are the types of chemical compounds that are obtained as result of electron sharing between bonded atoms. These compounds are generally formed between non-metallic elements and are also called molecular compounds.
Ionic compound:
Such compounds are producedby transfer of electrons from metallic to non-metallic atom. Ionic bond is formed due to existence of electronegativity difference between bonded atoms. Since metallic atoms are electropositive while non-metals are electronegative, metals lose electrons that are gained by non-metals.
Potassium and fluorine:
Potassium is metallic while fluorine is non-metallic in nature. So potassium loses its electron that is gained by fluorine. As a result, ions are formed and bond between potassium and fluorine is an ionic bond.
Aluminum and aluminum:
Aluminum is a metal. When bond is formed between two Al atoms, there occurs no difference in electronegativities of atoms and therefore ionic bond cannot be formed in this case.
Sulfur and oxygen:
Sulfur and oxygen are non-metals so bond formed between these two is covalent bond and not ionic bond.
Calcium and neon:
Calcium is a metal and it can lose electrons for bond formation. But neon is noble gas that is already having completely filled and stable octet in it. So no bond formation takes place between these two atoms.
Learn more:
Identification of ionic bonding:
What type of bond exists between phosphorus and chlorine?
Answer details:
Grade: High School
Subject: Chemistry
Chapter: Ionic and covalent compounds
Keywords: chemical bond, ionic bond, covalent bond, sulfur, oxygen, calcium, neon, aluminum, potassium, fluorine.
















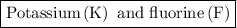 are more likely to form ionic bond.
are more likely to form ionic bond.




