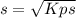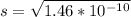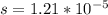Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 20:00, teacherpreacher
For the reaction c6h14(g) & longrightarrow; c6h6(g) + 4h2(g), δp(h2)/δt was found to be 2.5 x 10-2 atm/s, where δp(h2) is the change in pressure of hydrogen. determine δp(c6h14)/δt for this reaction at the same time.
Answers: 2

Chemistry, 22.06.2019 20:00, bbyjean9974
State one important difference between a physical change and a chemical change?
Answers: 1
Do you know the correct answer?
The ksp of zinc carbonate, znco3, is 1.46 × 10-10. calculate the molar solubility of this compound...
Questions in other subjects:


English, 17.04.2021 01:00


German, 17.04.2021 01:00

Mathematics, 17.04.2021 01:00


Mathematics, 17.04.2021 01:00

Mathematics, 17.04.2021 01:00

Mathematics, 17.04.2021 01:00

Mathematics, 17.04.2021 01:00


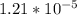
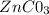 ⇒
⇒ 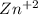 +
+ 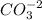
![Kps=[Zn^{+2} ]*[CO_{3} ^{-2} ]](/tpl/images/0353/9769/4395a.png)
![[Zn^{+2} ]=[CO_{3} ^{-2} ]=s](/tpl/images/0353/9769/64868.png)