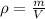Chemistry, 29.09.2019 09:20, elijahdouglass00
Iron has a density of 7.86 g/cm3. calculate the volume (in dl) of a piece of iron having a mass of 3.72 kg

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:30, KieraKimball
10-14. (a) when 100.0 ml of weak acid ha were titrated with 0.093 81 m naoh, 27.63 ml were required to reach the equivalence point. find the molarity of ha. (b) what is the formal concentration of a- at the equivalence point? (c) the ph at the equivalence point was 10.99. find pk. for ha. (d) what was the ph when only 19.47 ml of naoh had been added?
Answers: 1

Chemistry, 21.06.2019 21:10, danielahchf
How is 0.00235 expressed in proper scientific notation? a. 2.35 × 10-3 b. 0.235 × 10-2 c. 2.35 d. 2.35 × 103
Answers: 1

Do you know the correct answer?
Iron has a density of 7.86 g/cm3. calculate the volume (in dl) of a piece of iron having a mass of 3...
Questions in other subjects:


Mathematics, 12.03.2021 06:30

Mathematics, 12.03.2021 06:30





Mathematics, 12.03.2021 06:30

Mathematics, 12.03.2021 06:30