
Chemistry, 20.10.2019 11:20, proudmarinemom7186
The atomic symbol mc025-1.jpg represents potassium-40 (k-40), a radioactive isotope that has 19 protons and 21 neutrons. which atomic symbol could represent an isotope that results from the decay of k-40?
a) 40 ar
18
b) 40 k
18
c) 42 ca
20
d) 42 k
20

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 11:50, bellojamilet410
What substance has a mass of 9.5g and volume of 2.1cm^3
Answers: 2


Chemistry, 22.06.2019 14:00, jivsf
The two naturally occurring isotopes of chlorine are 35cl (34.969 amu, 75.77%) and 37cl (36.966 amu, 24.23%). the two naturally occurring isotopes of bromine are 79br (78.918 rm amu, 50.69%) and 81br (80.916 amu, 49.31%). chlorine and bromine combine to form bromine monochloride, brcl. 1. how many peaks will be present in a mass spectrum for brcl? the four combinations of molecule possible given these four isotopes are: 81br37cl, 81br35cl, 79br37cl, and 79br35cl. 2. what are the masses of the four different brcl molecules? express the masses using six significant figures, in decreasing numeric order (highest to lowest), separated by commas.
Answers: 3
Do you know the correct answer?
The atomic symbol mc025-1.jpg represents potassium-40 (k-40), a radioactive isotope that has 19 prot...
Questions in other subjects:

Arts, 14.12.2020 01:00

Spanish, 14.12.2020 01:00


Mathematics, 14.12.2020 01:00



English, 14.12.2020 01:00

Mathematics, 14.12.2020 01:00

Mathematics, 14.12.2020 01:00


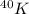 having 19 protons and 21 neutrons, the decay processes are beta-decay and electron capture.
having 19 protons and 21 neutrons, the decay processes are beta-decay and electron capture.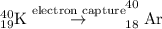
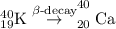
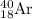 by electron capture. So, the correct option is A.
by electron capture. So, the correct option is A.



