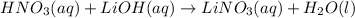Which of the statements below best describes the following reaction?
hno3(aq) + lioh(aq) → li...

Chemistry, 07.01.2020 09:31, yentel110306
Which of the statements below best describes the following reaction?
hno3(aq) + lioh(aq) → lino3(aq) + h2o(l)
select one:
a. nitric acid and lithium hydroxide produce lithium nitrate and water.
b. nitric acid and lithium hydroxide solutions react to produce lithium nitrate and water.
c. nitric acid and lithium hydroxide solutions react to produce lithium nitrate solution and water.
d. aqueous solutions of nitric acid and lithium hydroxide react to produce aqueous lithium nitrate and water.
e. an acid plus a base react to produce a salt and water.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, citlalli30
An atom of sodium-23 (atomic number = 11) has a positive charge of +1. give this information, how many electrons does it have? how many proteins and neutrons does this atom have
Answers: 2

Chemistry, 22.06.2019 09:40, cheesecake1919
Which diagram shows the correct way to represent an ionic compound of magnesium oxide?
Answers: 3

Chemistry, 22.06.2019 11:00, micro7909
Predict the products of the following acid-base reactions, and predict whether the equilibrium lies to the left or to the right of the reaction arrow. part ao2-(aq)+h2o(l)< => express your answer as part of a chemical equation. identify all of the phases in your answer. o2-(aq)+h2o(l) < => oh-(aq)+oh-(aq)part bpredict whether the equilibrium lies to the left or to the right of the equation in previous part. h2o is a stronger acid than oh–, so the equilibrium lies to the right. h2o is a weaker acid than oh–, so the equilibrium lies to the left. h2o is a stronger acid than oh–, so the equilibrium lies to the left. h2o is a weaker acid than oh–, so the equilibrium lies to the right. part cch3cooh(aq)+hs? (aq) < => express your answer as part of a chemical equation. identify all of the phases in your answer. ch3cooh(aq)+hs-(aq) < => h2s(aq)+c2h3o2-(aq)h2s(aq)+c2h3o2-( aq)part dpredict whether the equilibrium lies to the left or to the right of the equation in previous part. ch3cooh is a weaker acid than h2s, so the equilibrium lies to the right. ch3cooh is a weaker acid than h2s, so the equilibrium lies to the left. ch3cooh is a stronger acid than h2s, so the equilibrium lies to the right. ch3cooh is a stronger acid than h2s, so the equilibrium lies to the left. part eno2-(aq)+h2o(l) < => express your answer as part of a chemical equation. identify all of the phases in your answer. no2-(aq)+h2o(l) < => part fpredict whether the equilibrium lies to the left or to the right of the equation in previous part. hno2 is a stronger acid than h2o, so the equilibrium lies to the right. hno2 is a weaker acid than h2o, so the equilibrium lies to the left. hno2 is a stronger acid than h2o, so the equilibrium lies to the left. hno2 is a weaker acid than h2o, so the equilibrium lies to the right.
Answers: 1

Chemistry, 22.06.2019 13:30, hdhtvthjr
Which of the following natural processes is most likely to support the formation of an underwater sinkhole? a pollution buildup from deposited minerals b limestone cave collapsing due to changes in sea level c erosion of large amounts of sand moved by ocean waves d oxidation of rock formed by chemical weathering
Answers: 1
Do you know the correct answer?
Questions in other subjects:

Health, 21.04.2020 00:54

Mathematics, 21.04.2020 00:54

Mathematics, 21.04.2020 00:54


Mathematics, 21.04.2020 00:54

Mathematics, 21.04.2020 00:54


Mathematics, 21.04.2020 00:54


Mathematics, 21.04.2020 00:54