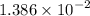(6.93 x 10 1 ) (2.0 x 10 -4 ) =
1.386 -2
13.86 x 10 -3
1.386 x 10 -2
...

Chemistry, 29.01.2020 16:57, lilpeepxliltracy
(6.93 x 10 1 ) (2.0 x 10 -4 ) =
1.386 -2
13.86 x 10 -3
1.386 x 10 -2
1.4 x 10 -2

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, sotoamerica0814
98 ! and brainliest plz ! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 06:00, nikejose11
An alkaline battery produces electrical energy according to the following equation. zn(s) + 2 mno2(s) + h2o(l) zn(oh)2(s) + mn2o3(s) (a) determine the limiting reactant if 17.5 g zn and 31.0 g mno2 are used. (type your answer using the format ch4 for ch4.) (b) determine the mass of zn(oh)2 produced. _ g
Answers: 3


Chemistry, 22.06.2019 12:40, valenzueladomipay09u
How does concentration affect reaction rate
Answers: 2
Do you know the correct answer?
Questions in other subjects:

Mathematics, 06.10.2020 23:01

Mathematics, 06.10.2020 23:01



Chemistry, 06.10.2020 23:01

Mathematics, 06.10.2020 23:01