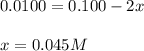10.0 ml of a 0.100 mol l–1 solution of a metal ion m2+ is mixed with 10.0 ml of a 0.100 mol l–1 solution of a substance l. the following equilibrium is established:
m2+(aq) + 2l(aq) picture ml22+(aq)
at equilibrium the concentration of l is found to be 0.0100 mol l–1. what is the equilibrium concentration of ml22+, in mol l–1?
someone me

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:00, pettygirl13
Describe the particles of all three phases of matter in the root beer float. (how are the particles arranged and moving? )
Answers: 3


Do you know the correct answer?
10.0 ml of a 0.100 mol l–1 solution of a metal ion m2+ is mixed with 10.0 ml of a 0.100 mol l–1 solu...
Questions in other subjects:





History, 24.02.2021 21:20

Chemistry, 24.02.2021 21:20

Mathematics, 24.02.2021 21:20


Mathematics, 24.02.2021 21:20


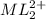 at equilibrium is 0.045 M.
at equilibrium is 0.045 M.![[M^{2+}]_{initial}=0.100M](/tpl/images/0204/7051/8debc.png)
![[L]_{initial}=0.100M](/tpl/images/0204/7051/9e7cd.png)
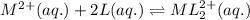
![[L]_{eqllm}=0.0100M](/tpl/images/0204/7051/18682.png)