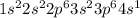Two representations of the distribution of electrons in the potassium atom are shown.
mo...

Two representations of the distribution of electrons in the potassium atom are shown.
model 1 model 2
1s22s22p63s23p64s1
which model is useful in showing the reactivity of potassium?
neither model is able to show the reactivity of potassium.
model 1 because it shows an electron in the 4s orbital.
model 2 because it shows an electron in the outermost ring.
both models because each shows an electron in the outermost shell.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 02:30, brittanysanders
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2


Chemistry, 22.06.2019 18:00, kingamir
Answer asap need it by wednesday morning carry out the following calculations on ph and ka of from data. i. calculate the ph of 0.02m hcl ii. calculate the ph of 0.036m naoh iii. calculate the ph of 0.36m ca(oh)2 iv. calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 v. calculate ka for weak acid ha which has a ph of 3.65 at 0.30m concentration vi. calculate the ka of a solution made by mixing 15.0 cm3 0.2m ha and 60.0 cm3 0.31m a-. [ph= 3.80] vii. calculate the ph of a solution made by mixing 15.0 cm3 0.1m naoh and 35.0 cm3 0.2m hcooh. [ka = 1.82 x 10-4 m]
Answers: 1
Do you know the correct answer?
Questions in other subjects:



Physics, 16.04.2021 20:30


English, 16.04.2021 20:30

Mathematics, 16.04.2021 20:30

Mathematics, 16.04.2021 20:30


Health, 16.04.2021 20:30