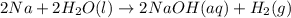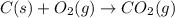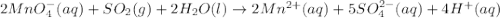Answers: 1
Other questions on the subject: Chemistry

Chemistry, 23.06.2019 08:00, oopsorry
Determine the number of moles of air present in 1.35 l at 750 torr and 17.0°c. which equation should you use? n=pv/rt what is the number of moles present? ⇒ 0.056 mol a sample of n2 gas occupying 800.0 ml at 20.0°c is chilled on ice to 0.00°c. if the pressure also drops from 1.50 atm to 1.20 atm, what is the final volume of the gas? which equation should you use? v2= p1v1t2/p2t1 what is the final volume of the gas? ⇒ 932 ml these are the answers
Answers: 1

Chemistry, 23.06.2019 10:30, thompsonjodi9638
Chemical bonds result from the interaction of the from two or more atoms. a. protons b. electrons c. neutrons d. nuclei
Answers: 2

Chemistry, 23.06.2019 11:20, bobthebattlebot
The chemical composition of soil varies with depth. an article in communications in soil science and plant analysis describes chemical analyses of soil taken from a farm in western australia. fifty specimens were each taken at depths 50 and 250 cm. at a depth of 50 cm, the average no3 concentration (in mg/l) was 88.5 with a standard deviation of 49.4. at a depth of 250 cm, the average concentration was 110.6 with a standard deviation of 51.5. find a 95% confidence interval for the difference in no3 concentrations at the two depths.
Answers: 1
Do you know the correct answer?
What are the oxidizing agents, and the reducing agents for 2na(aq)+2h2o(l)→2naoh(aq)+h2(g)
c(s...
c(s...
Questions in other subjects:


Spanish, 04.07.2019 08:30

Mathematics, 04.07.2019 08:30

Mathematics, 04.07.2019 08:30


Arts, 04.07.2019 08:30

Computers and Technology, 04.07.2019 08:30

History, 04.07.2019 08:30

History, 04.07.2019 08:30