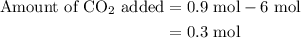Chemistry, 30.01.2020 05:57, Queiao4088
An equilibrium mixture contains 0.600 mol of each of the products (carbon dioxide and hydrogen gas) and 0.200 mol of each of the reactants (carbon monoxide and water vapor) in a 1.00-l container. how many moles of carbon dioxide would have to be added at constant temperature and volume to increase the amount of carbon monoxide to 0.300 mol once equilibrium has been reestablished?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, kingteron5870
Ionic compounds are made of ions, and yet the overall charge of an ionic compound is neutral. why?
Answers: 1

Chemistry, 22.06.2019 01:00, jescanarias22
Which statement correctly describes potassium iodide, ki? there is a one-to-one ratio of potassium ions to iodide ions. potassium gains electrons and iodine loses electrons during the reaction. the lattice is held together by potassium anions and iodide cations.
Answers: 1


Do you know the correct answer?
An equilibrium mixture contains 0.600 mol of each of the products (carbon dioxide and hydrogen gas)...
Questions in other subjects:

Spanish, 30.07.2019 14:30


Social Studies, 30.07.2019 14:30

History, 30.07.2019 14:30


Social Studies, 30.07.2019 14:30


Social Studies, 30.07.2019 14:30


Business, 30.07.2019 14:30


 of
of  are added so as to increase the amount of carbon monoxide to 0.3 mol.
are added so as to increase the amount of carbon monoxide to 0.3 mol.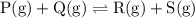
![{\text{K}}=\dfrac{{\left[ {\text{R}} \right]\left[ {\text{S}}\right]}}{{\left[{\text{P}}\right]\left[ {\text{Q}} \right]}}](/tpl/images/0484/7673/9b899.png)
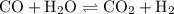
![{\text{K = }}\dfrac{{\left[ {{\text{C}}{{\text{O}}_{\text{2}}}} \right]\left[{{{\text{H}}_{\text{2}}}} \right]}}{{\left[ {{\text{CO}}}\right]\left[{{{\text{H}}_2}{\text{O}}} \right]}}](/tpl/images/0484/7673/6dcad.png) .......(1)
.......(1)![\left[{{\text{C}}{{\text{O}}_{\text{2}}}}\right]](/tpl/images/0484/7673/9014c.png) is the concentration of carbon dioxide.
is the concentration of carbon dioxide.
![\left[{{{\text{H}}_{\text{2}}}}\right]](/tpl/images/0484/7673/340fe.png) is the concentration of hydrogen.
is the concentration of hydrogen.
![\left[{{\text{CO}}}\right]](/tpl/images/0484/7673/d6da7.png) is the concentration of carbon monoxide.
is the concentration of carbon monoxide.
![\left[{{{\text{H}}_2}{\text{O}}}\right]](/tpl/images/0484/7673/62a9e.png) is the concentration of water.
is the concentration of water.
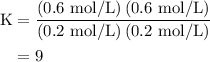
![\left[{{\text{C}}{{\text{O}}_{\text{2}}}}\right]=\dfrac{{{\text{K}}\left( {\left[{{\text{CO}}} \right]\left[{{{\text{H}}_2}{\text{O}}}\right]}\right)}}{{\left[{{{\text{H}}_{\text{2}}}} \right]}}](/tpl/images/0484/7673/f6940.png) ......(2)
......(2)![\begin{aligned}\left[ {{\text{C}}{{\text{O}}_{\text{2}}}}\right]&= \frac{{{\text{9}}\left( {{\text{0}}{\text{.3 mol/L}}}\right)\left({{\text{0}}{\text{.2 mol/L}}}\right)}}{{{\text{0}}{\text{.6 mol/L}}}}\\&= 0.{\text{9 mol/L}}\\\end{aligned}](/tpl/images/0484/7673/95ec6.png)