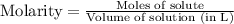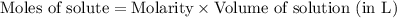Which of the following operations yields the number of moles of solute?
a. molarity x liters...

Chemistry, 25.11.2019 13:31, damiangibson2
Which of the following operations yields the number of moles of solute?
a. molarity x liters of solution
b. molarity x moles of solution
c. molarity x mass of solution
d. moles of solution /volume ofsolution?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:40, timmonskids6027
Consider the elements bromine and chlorine; which elements has a larger ionic radius ?
Answers: 1

Chemistry, 22.06.2019 10:10, jojomgarcia01
When water dissociates, each water molecule splits into a hydroxide ion and a) h 3 o + b) a hydrogen atom c) a hydrogen ion d) h 2 o e) oh —
Answers: 2

Chemistry, 22.06.2019 13:00, netflixacc0107
Amixture with the same composition throughout is!
Answers: 1

Chemistry, 22.06.2019 14:00, IdkHowToDoMath
What term describes technology that operates on an atomic level
Answers: 2
Do you know the correct answer?
Questions in other subjects:


English, 03.02.2020 12:56







Physics, 03.02.2020 12:56