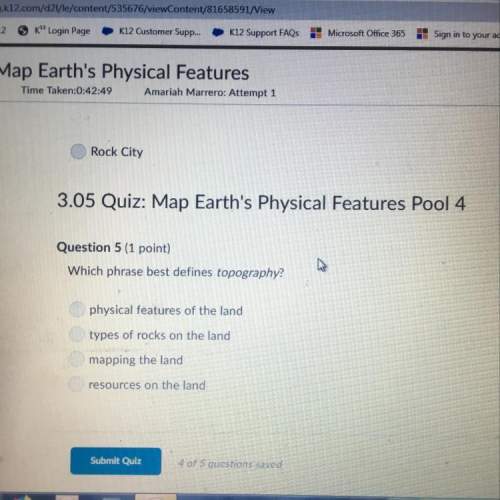
Calculate the number of moles in 75.0g of dinitrogen trioxide
...

Chemistry, 11.10.2019 01:30, brittanyfox411
Calculate the number of moles in 75.0g of dinitrogen trioxide


Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:30, minstcordell4115
Covalent network solids typically have melting points and boiling points. the chemical formula of a network solid indicates in the molecule.
Answers: 3

Chemistry, 22.06.2019 05:30, maddyjones4172
Which of the following two events occur to create a sea breeze? select all that apply. warm air rises on the ocean and moves toward the land to cool warm air rises on land and moves toward the ocean to cool cool air moves from the ocean to be warmed by the land cool air moves from the land to be warmed by the ocean
Answers: 3

Chemistry, 22.06.2019 09:00, SilverTheAmarok
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 2

Chemistry, 22.06.2019 11:00, 21villalobosjabez
Which type of fossil does this image depict?
Answers: 1
Do you know the correct answer?
Questions in other subjects:

Mathematics, 28.08.2019 16:30

History, 28.08.2019 16:30


Mathematics, 28.08.2019 16:30





Geography, 28.08.2019 16:30