
Chemistry, 05.10.2019 17:50, gonzmari457
1. when an acid reacts with a base, what compounds are formed? (1 point) a salt only water only metal oxides only a salt and water2. the formula of the hydrogen ion is often written as (1 point) h2o+ oh+ h+ h4n+ 3. what is an acid according to arrhenius? (1 point) a substance that ionizes to yield protons in aqueous solution a substance that is a hydrogen ion donor a substance that accepts an electron pair a substance that is a hydrogen ion acceptor 4. what is transferred between a conjugate acidbase pair? (1 point) an electron a hydroxide ion a hydrogen ion a hydronium ion 5. which of the following represents a brønstedlowry conjugate acidbase pair? (1 point) so3 2and so2 co3 2and co h3o and h2 nh4 + and nh3

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, bbombard21
Select the atomic models that belong to the same element
Answers: 2



Chemistry, 23.06.2019 04:00, ayoismeisjjjjuan
How many liters of water can be produced from 5.0liters of butane gas at stp, assuming excess oxygen? c4h10(g) + 02(g) → co2 (e) + h2o (g)
Answers: 2
Do you know the correct answer?
1. when an acid reacts with a base, what compounds are formed? (1 point) a salt only water only met...
Questions in other subjects:


Arts, 14.01.2021 22:40

Chemistry, 14.01.2021 22:40







Spanish, 14.01.2021 22:50



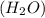 .
.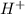 .
.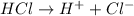
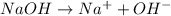
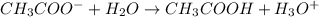
 accepts a proton and thus act as a base and the corresponding
accepts a proton and thus act as a base and the corresponding 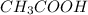 is its conjugate acid.
is its conjugate acid. and
and 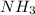
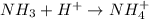
 is a acid which accepts proton and thus acts as base to form conjugate acid
is a acid which accepts proton and thus acts as base to form conjugate acid 



