

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:50, JuniperGalaxy
Consider the equilibrium system: 2icl(s) ⇄ i2(s) + cl2(g) which of the following changes will increase the total amount of of cl2 that can be produced? all of the listed answers are correct decreasing the volume of the container removing the cl2 as it is formed adding more icl(s) removing some of the i2(s)
Answers: 1


Chemistry, 22.06.2019 13:00, carlinryan
16. why must the number of electrons lost equal the number of electrons gained in every redox reaction? use 3 – 4 sentences in your own words to address this question. 18. what type of radiation is emitted when chromium-51 decays into manganese-51? show the nuclear equation that leads you to this answer. 19. a radioactive nucleus alpha decays to yield a sodium-24 nucleus in 14.8 hours. what was the identity of the original nucleus? show the nuclear equation that leads you to this answer.
Answers: 2

Chemistry, 22.06.2019 20:30, kittybatch345
Is a chemical message sent by another individual.
Answers: 1
Do you know the correct answer?
Identify the strongest attractive forces between the particles of each of the following a. ch3oh b....
Questions in other subjects:

Biology, 12.03.2021 19:40




Mathematics, 12.03.2021 19:40


Mathematics, 12.03.2021 19:40

Chemistry, 12.03.2021 19:40

Business, 12.03.2021 19:40

Chemistry, 12.03.2021 19:40


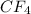 has four polar covalent bonds but the over all molecule is nonpolar as the dipole moment of one C-F bond is canceled by its opposite C-F bond as the molecule is tetrahedral. Being nonpolar, the molecule has london dispersion forces only.
has four polar covalent bonds but the over all molecule is nonpolar as the dipole moment of one C-F bond is canceled by its opposite C-F bond as the molecule is tetrahedral. Being nonpolar, the molecule has london dispersion forces only.



