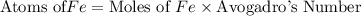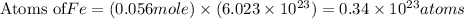Chemistry, 05.10.2019 19:10, sammielucero
How many iron atoms would be produced by 4.5g of fe2o3 reacting eith an excess of magnesium

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, caeyanij
When svante arrhenius first proposed his acid-base theory, he was a doctoral candidate. his professors thought his ideas were unfounded. within a decade, the arrhenius theory of acid-base was widely accepted and praised within the scientific world. arrhenius defined acids as compounds having ionizable hydrogen and bases as compounds with ionizable a) barium. b) hydronium. c) hydroxide. d) oxygen.
Answers: 3


Chemistry, 22.06.2019 08:30, masontdavis
Draw the skeletal structures of two different molecules that are each made of 5 carbon atoms and 12 hydrogen atoms.
Answers: 1

Chemistry, 22.06.2019 10:30, zayam1626
Great amounts of electromagnetic energy from our sun and other bodies in space travel through space. which is a logical conclusion about these electromagnetic waves? their energy must be very their frequency must be very low these waves can travel without a medium they only travel through a vacuum of space
Answers: 2
Do you know the correct answer?
How many iron atoms would be produced by 4.5g of fe2o3 reacting eith an excess of magnesium...
Questions in other subjects:

Health, 17.10.2019 12:30




Biology, 17.10.2019 12:30


History, 17.10.2019 12:30


Mathematics, 17.10.2019 12:30

Physics, 17.10.2019 12:30


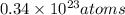
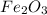 = 4.5 g
= 4.5 g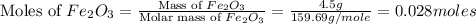
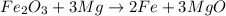
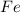
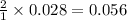 moles of
moles of