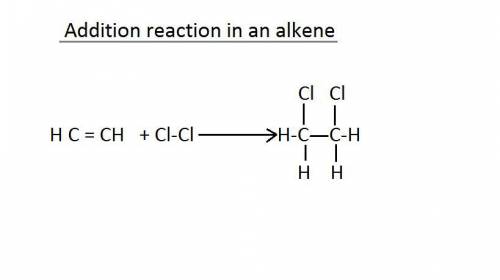
In an addition reaction, which bond of the reactant is broken?
a. carbon carbon single bond...

Chemistry, 02.10.2019 06:30, mimireds5419
In an addition reaction, which bond of the reactant is broken?
a. carbon carbon single bond
c. carbon carbon double bond
b. carbon hydrogen single bond
d. carbon hydrogen double bondin chemistry, a hydrogen bond is a type of attractive intermolecular force that exists between two partial electric charges of opposite polarity. [ although stronger than most other intermolecular forces, the hydrogen bond is much weaker than both the ionic bond and the covalent bond. ]

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:40, trinityanne1738
Asolid that forms and separates from a liquid mixture is called
Answers: 2

Chemistry, 22.06.2019 21:00, cxttiemsp021
The rate constant for the reaction below is 6.2 x 10−5 mol l−1 s −1. if the initial concentration of a is 0.0500 m, what is its concentration after 115 s?
Answers: 1

Chemistry, 22.06.2019 22:30, darceline1574
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3
Do you know the correct answer?
Questions in other subjects:

Biology, 09.11.2020 21:40


Health, 09.11.2020 21:40

Law, 09.11.2020 21:40




Mathematics, 09.11.2020 21:40


Mathematics, 09.11.2020 21:40