Given the balanced equation representing a reaction:
zn(s) + h2so4(aq) → znso4(aq) + h2...

Chemistry, 03.11.2019 18:31, naomicervero
Given the balanced equation representing a reaction:
zn(s) + h2so4(aq) → znso4(aq) + h2(g)
which type of reaction is represented by this equation?
1.
decomposition
2.
double replacement
3.
single replacement
4.
synthesis

Answers: 2
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 17:00, brownvester44
Astable electron arrangement for an atom is one that does not easily change. how is this arrangement arrived at? a. valence electrons are transferred or shared to create a full outer shell of electrons. b. valence electrons are discarded into space to create a full outer shell of electrons. c. protons (positive charge) pair with valence electrons (negative charge) to create a strong bond. d. outer shells with valence electrons are transferred or shared.
Answers: 2

Chemistry, 22.06.2019 18:00, kingamir
Answer asap need it by wednesday morning carry out the following calculations on ph and ka of from data. i. calculate the ph of 0.02m hcl ii. calculate the ph of 0.036m naoh iii. calculate the ph of 0.36m ca(oh)2 iv. calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 v. calculate ka for weak acid ha which has a ph of 3.65 at 0.30m concentration vi. calculate the ka of a solution made by mixing 15.0 cm3 0.2m ha and 60.0 cm3 0.31m a-. [ph= 3.80] vii. calculate the ph of a solution made by mixing 15.0 cm3 0.1m naoh and 35.0 cm3 0.2m hcooh. [ka = 1.82 x 10-4 m]
Answers: 1
Do you know the correct answer?
Questions in other subjects:

English, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

English, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01


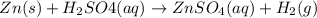 is a single replacement reaction.
is a single replacement reaction.